Author: Harold Chen, MD, MS, FAAP, FACMG; Chief Editor: Bruce Buehler, MD
Marco Fraccaro first described achondrogenesis in 1952.[1] He used the term to describe a stillborn female with severe micromelia and marked histological cartilage changes. The term was later used to characterize the most severe forms of chondrodysplasia in humans, which were invariably lethal before or shortly after birth. By the 1970s, researchers concluded that achondrogenesis was a heterogeneous group of chondrodysplasias lethal to neonates; achondrogenesis type I (Fraccaro-Houston-Harris type) and type II (Langer-Saldino type) were distinguished on the basis of radiological and histological criteria.
See the image below.
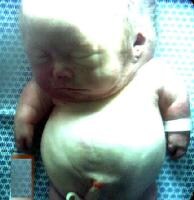 An infant with achondrogenesis type II. Note the disproportionately large head, large and prominent forehead, flat facial plane, flat nasal bridge, small nose with severely anteverted nostrils, micrognathia, extremely short neck, short and flared thorax, protuberant abdomen, and extremely short upper extremities.
An infant with achondrogenesis type II. Note the disproportionately large head, large and prominent forehead, flat facial plane, flat nasal bridge, small nose with severely anteverted nostrils, micrognathia, extremely short neck, short and flared thorax, protuberant abdomen, and extremely short upper extremities.
In 1983, a new radiological classification of achondrogenesis (types I-IV) by Whitley and Gorlin was adopted in the McKusick catalog.[2] According to this classification, type I and type II have the same femoral cylinder index (CIfemur; calculated as length of femur divided by width of femur) range (1-2.8). Both types have crenated ilia and stellate long bones. Multiple rib fractures are characteristic of type I but not type II. Type III has nonfractured ribs, halberd ilia, mushroom-stem long bones, and a CIfemur of 2.8-4.9. Type IV has nonfractured ribs, sculpted ilia, well-developed long bones, and a CIfemur of 4.9-8. This radiological classification based on the CIfemur was later abandoned. Researchers suggested that achondrogenesis type III probably corresponds to type II and that type IV probably corresponds to mild type II (hypochondrogenesis).
See the images below.
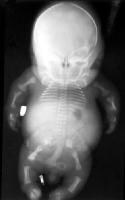 This posteroanterior (PA) view radiograph of an infant with achondrogenesis type II shows the relatively large calvaria with normal cranial ossification, short and flared thorax, bell-shaped cage and shorter ribs without fractures, relatively well ossified iliac bone with long crescent-shaped medial and inferior margins, and short tubular bones. The sacrum, pubis, and ischium are not visible.
This posteroanterior (PA) view radiograph of an infant with achondrogenesis type II shows the relatively large calvaria with normal cranial ossification, short and flared thorax, bell-shaped cage and shorter ribs without fractures, relatively well ossified iliac bone with long crescent-shaped medial and inferior margins, and short tubular bones. The sacrum, pubis, and ischium are not visible.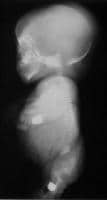 Lateral view radiograph of an infant with achondrogenesis type II. Note the relatively large head with a normal cranial ossification and enlarged fontanelles, short ribs, absent sternal ossification, ossification only in anterior parts of the vertebral bodies, and short and curved femora.
Lateral view radiograph of an infant with achondrogenesis type II. Note the relatively large head with a normal cranial ossification and enlarged fontanelles, short ribs, absent sternal ossification, ossification only in anterior parts of the vertebral bodies, and short and curved femora.
In the late 1980s, structural mutations in collagen II were shown to cause achondrogenesis type II, which thus constitutes the severe end of the spectrum of collagen II chondrodysplasias. Achondrogenesis type I was subdivided further in 1988 on the basis of convincing histological criteria. It was subdivided into type IA, which has apparently normal cartilage matrix but inclusions in chondrocytes, and type IB, which has an abnormal cartilage matrix. Classification of type IB as a separate group has been confirmed by the discovery of its association with mutations in the diastrophic dysplasia sulfate transporter (DDST) gene, making it allelic with diastrophic dysplasia.
Currently, 3 variants of achondrogenesis have been defined based on radiologic and histopathologic features: type IA (Houston-Harris), type IB (Parenti-Fraccaro), and type II (Langer-Saldino). Achondrogenesis IA appears to be autosomal recessive, but the mutant gene is still unknown. Type IB is caused by recessive mutations of the diastrophic dysplasia sulfate transporter gene (SLC26A2), and type II is caused by autosomal dominant mutations of the type II collagen gene (COL2A1).
Achondrogenesis II results from heterozygosity for a new dominant mutation in theCOL2A1 gene at the chromosomal locus 12q8.11–q13.2. Intramolecular heterogeneity has been recognized, and genotype–phenotype correlations have been demonstrated.[3]
Several heritable osteochondrodysplasias have now been recognized as members of the family of type II collagen disorders, all of which result from dominant mutations in the COL2A1 gene.[4, 5, 3] Phenotypes within this group range from severe lethal dwarfism at birth to relatively mild conditions with precocious osteoarthrosis and little or no skeletal growth abnormality. Achondrogenesis II-hypochondrogenesis and lethal spondyloepiphyseal dysplasia congenita (SEDC) represent the more severe end of the spectrum.[6] These entities are characterized by severe disproportionate short stature of prenatal onset. The distinction between these phenotypes is mainly based on clinical, radiographic, and morphological features but considerable phenotypic overlap often hampers proper classification.
Mutations within the COL2A1 gene also cause hypochondrogenesis (OMIM 200610), spondyloepiphyseal dysplasia (SED) congenita (OMIM 183900), SED Namaqualand type (OMIM 142670), mild SED with precocious osteoarthritis, spondyloepimetaphyseal dysplasia Strudwick type (OMIM 184250), Kniest dysplasia (OMIM 156550), multiple epiphyseal dysplasia with myopia and conductive deafness, spondyloperipheral dysplasia (OMIM 271700), and Stickler dysplasia type I (OMIM 108300).[7]
Pathophysiology
- A series of mutations in the DDST gene has been identified in patients with achondrogenesis type IB. Homozygosity or compound heterozygosity for these mutations, which leads to premature stop codons or structural mutations in transmembrane domains, is associated with achondrogenesis type IB. Extracellular loops or cytoplasmic tail mutations or low messenger RNA (mRNA) levels, which cause regulatory mutation, usually result in atelosteogenesis type II or diastrophic dysplasia with less severe phenotypes. Chondrocytes and skin fibroblasts cultured from patients with type IB are unable to incorporate exogenous sulfate.
- Different mutations in the gene that encodes type II collagen (COL2A1) cause achondrogenesis type II as well as other type II collagenopathies (eg, spondyloepiphyseal dysplasias, hypochondrogenesis). Type II has a single base change, substituting serine for glycine in the type II procollagen gene of the alpha 1(II) chain. This disrupts the triple helix formation, leading to a paucity of type II collagen in the cartilage matrix. Epiphyseal cartilage lacks type II collagen. It is replaced by type I and type III collagens, which are not normally produced by chondrocytes. Differentiated chondrocytes do not express type II collagen. In addition to skeletal abnormalities, severe pulmonary hypoplasia, thought to be directly related to the underlying pathology in collagen expression, is associated with achondrogenesis.
- Type II achondrogenesis/hypochondrogenesis (Whitley and Gorlin prototype IV) has immunohistologic findings that demonstrate apparent abnormal intracellular accumulation of type II collagen within vacuolar structures of chondrocytes. This suggests the presence of abnormal, poorly secreted type II collagen. Molecular defects of type II collagen and new dominant mutations account for the observed phenotype.
Epidemiology
Frequency
United States
- Lethal achondrogenesis types I and II are both rare.
- Their respective frequencies are unknown; however, the overall frequency has been estimated at 1 in 40,000 births.
Mortality/Morbidity
- Achondrogenesis type I results in stillbirth more frequently than type II.
- Babies with achondrogenesis type I who are not stillborn typically have a shorter gestation and survive for a shorter time than those with type II. They are also smaller with much shorter limbs, which supports the general view that type I is the more severe form.
Race
- Achondrogenesis has no racial predilection.
Sex
- Males and females are equally affected.
Age
- Achondrogenesis is detected prenatally or at birth because of typical clinical, radiological, histological, and molecular findings.